 
Pressure, like temperature and volume, is a basic thermodynamic parameter, but it exhibits unique advantages in finding new materials and stabilizing unexpected stoichiometric compounds with new oxidation states. This is because compounds with new oxidation states usually contain new types of chemical bonding and exhibit interesting physical and chemical properties. On the other hand, the preparation of compounds with new oxidation states is a rather attractive topic in condense-mater physics and chemistry. Thus, the number of the valence electrons of an element is closely related to the oxidation state in its compounds. However, the inner-shell electrons or outer empty orbital are not involved in chemical bonding. In general, atoms react with other atoms by losing, obtaining, or sharing their valence electrons. 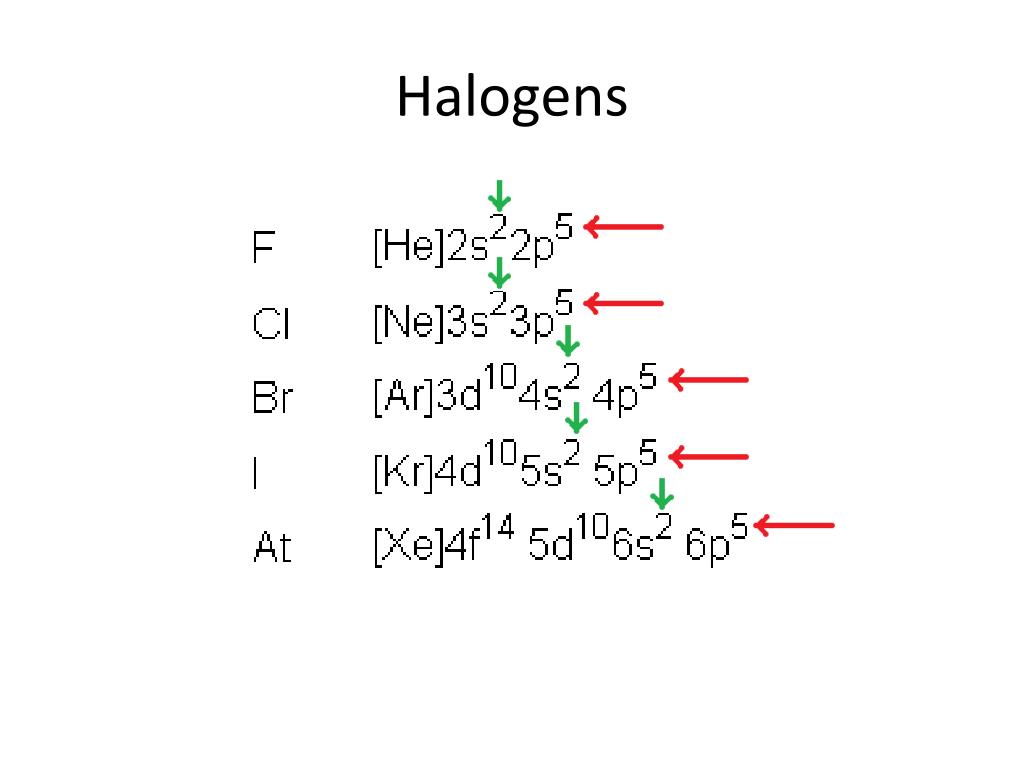
Personal outlook on the challenge and opportunity in this field are proposed in the conclusion.Īt ambient conditions, the valence electrons of an atom dominate the chemical properties, rooted in the well-accepted atomic shell structure. The aim of this review is to provide an up-to-date research progress on the chemical bonding with inner-shell electrons or outer empty orbital, abnormal interatomic charge transfer, hypervalent compounds, and chemical reactivity of noble gases. Here, we mainly focus on the recent advances in high-pressure new chemistry including novel chemical bonding and new oxidation state, identified by first-principles swarm intelligence structural search calculations. 
More recently, pressure stabilized a series of unconventional stoichiometric compounds with new oxidation states, in which the inner-shell electrons or outer empty orbital become chemically active. Pressure, as a fundamental thermodynamic variable, plays an important role in the preparation of new materials. It has long been recognized that the valence electrons of an atom dominate the chemical properties, while the inner-shell electrons or outer empty orbital do not participate in chemical reactions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
